Two-Year BIOPACT Randomized Controlled Trial (RCT) Analysis Demonstrates “Persistent Excellence” for Low-Profile Passeo-18 Lux DCB Latest Results of Head-to-Head Non-Inferiority Study Shows Continued Equivalent Safety and Efficacy of Passeo-18 Lux Drug-Coated Balloon (DCB) to In.Pact Admiral DCB in Treatment of Femoropopliteal Lesions
BIOTRONIK announced the presentation of two-year results from the investigator-initiated BIOPACT RCT by Principal Investigator Dr. Koen Deloose at the Paris Vascular Insights 2023 congress. The randomized controlled non-inferiority trial evaluated the safety and efficacy of the Passeo®-18 Lux® drug-coated balloon (DCB) catheter compared to the In.Pact Admiral DCB (Medtronic), and showed excellent results for both balloons through 24 months.1
The prospective, multicenter, core-lab adjudicated non-inferiority trial enrolled 302 patients in Austria, Belgium, France and Switzerland with Rutherford Class 2-4 disease. Patients were randomized 1:1 to either the Passeo-18 Lux DCB or In.Pact Admiral DCB for the treatment of stenotic, non-stented restenotic or occlusive lesions in the femoropopliteal artery. Baseline patient and lesion characteristics were comparable across both arms.
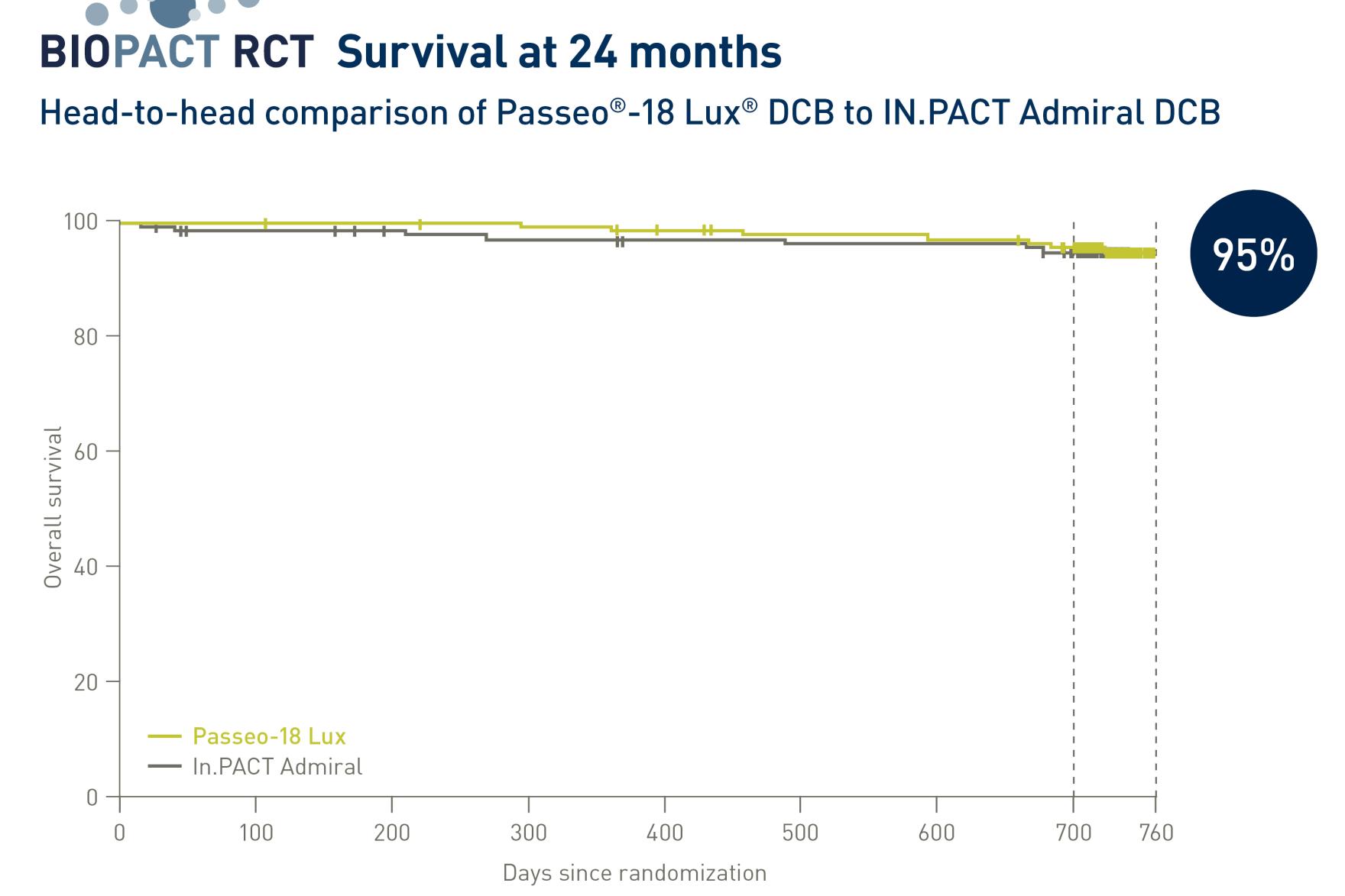
As presented at Paris Vascular Insights in November 2023, the rate of clinically driven target lesion revascularization (CD-TLR) for the Passeo-18 Lux DCB was noninferior to the In.Pact Admiral DCB at 12 months, which was maintained at 24 months (93.3%, Passeo-18 Lux DCB versus 94.6%, In.Pact Admiral DCB at 24 months; p-value = 0.0048).2
Survival was also equivalent at 24 months, with a 95.0% rate of overall survival for both devices, and there were no major target limb amputations in either arm.
“The 2-year results of the BIOPACT RCT have continued to demonstrate the noninferiority of the safety and efficacy of the Passeo-18 Lux DCB to the IN.PACT Admiral DCB, and they show persistent excellence of both devices,” said Dr. Koen Deloose, Head of the Department of Vascular Surgery, AZ Sint Blasius Hospital in Dendermonde, Belgium. “This study is critically needed to fill the gap of head-to-head data for peripheral DCBs to keep the most effective treatments accessible to our patients.”
The Passeo-18 Lux DCB has a paclitaxel dose of 3.0 µg/mm2 and uses a Butyryl-tri-hexyl citrate excipient (In.Pact Admiral DCB: 3.5 µg/mm2 and urea excipient).3 The lower-profile Passeo-18 Lux DCB is on an 0.018” platform and is 4-French compatible for diameters of 2.0 to 4.0 mm and 5-French compatible for diameters of 5.0 to 7.0 mm.3 In comparison, the In.Pact Admiral DCB is 5-French compatible only for its 4.0 mm diameter, and is 6-French or 7-French compatible for its diameters of 5.0 to 7.0 mm.4
“We are delighted to see confirmation of the continued performance and safety of our Passeo-18 Lux DCB in this important RCT, which will support our access in key new markets,” said Stuart Perks, Vice President Marketing Vascular Intervention at BIOTRONIK.
The BIOPACT RCT study will continue collecting follow-up data at 36, 48 and 60 months, which will be shared upon completion of each follow-up period.
-END-
References:
1. Deloose K. The head-to-head Passeo-18 Lux vs IN.PACT Admiral BIOPACT RCT: latest release of the 24 month outcomes. Presented at: Paris Vascular Insights 2023; November 8-10, 2023; Paris, France.
2. Deloose K, et al. Methodology of the BIOPACT RCT, a Multi-center, Randomized, Non-inferiority Trial Evaluating Safety and Efficacy of Passeo-18 Lux Drug-Coated Balloon (DCB) of Biotronik Compared to the Medtronic IN.PACT Admiral DCB in the Treatment of Subjects with Lesions of the Femoropopliteal Artery. Cardiovasc Intervent Radiol. 2022 Dec;45(12):1855-1859. doi: 10.1007/s00270-022-03259-z. Epub 2022 Sep 4. PMID: 36058996.
3. BIOTRONIK data on file.
Disclaimer:
Passeo-18 Lux paclitaxel-releasing PTA balloon catheter is not available for sale or use in the United States.
Passeo and Lux are trademarks or registered trademarks of the BIOTRONIK Group of Companies. All other trademarks or registered trademarks cited herein are the property of their respective owners.
For more information, please visit: Passeo-18 Lux DCB
About BIOTRONIK:
At BIOTRONIK, patient well-being is our top priority and has been for 60 years. BIOTRONIK is a leading global medical technology company with products and services that save and improve the lives of millions suffering from heart and blood vessel diseases as well as chronic pain. Driven by a purpose to perfectly match technology with the human body, we are dedicated innovators who develop trusted cardiovascular, endovascular and neuromodulation solutions. BIOTRONIK is headquartered in Berlin, Germany, and is represented in over 100 countries.