BIOLUX P-III BENELUX All-Comers Registry Demonstrates Safety and Efficacy of BIOTRONIK’s Drug-Coated Balloon in Isolated Popliteal Lesions at 24 Months First Dedicated Registry to Investigate DCB Performance in Isolated Popliteal Artery Lesions Shows Passeo-18 Lux DCB as Beneficial Treatment for Challenging Anatomical Segment
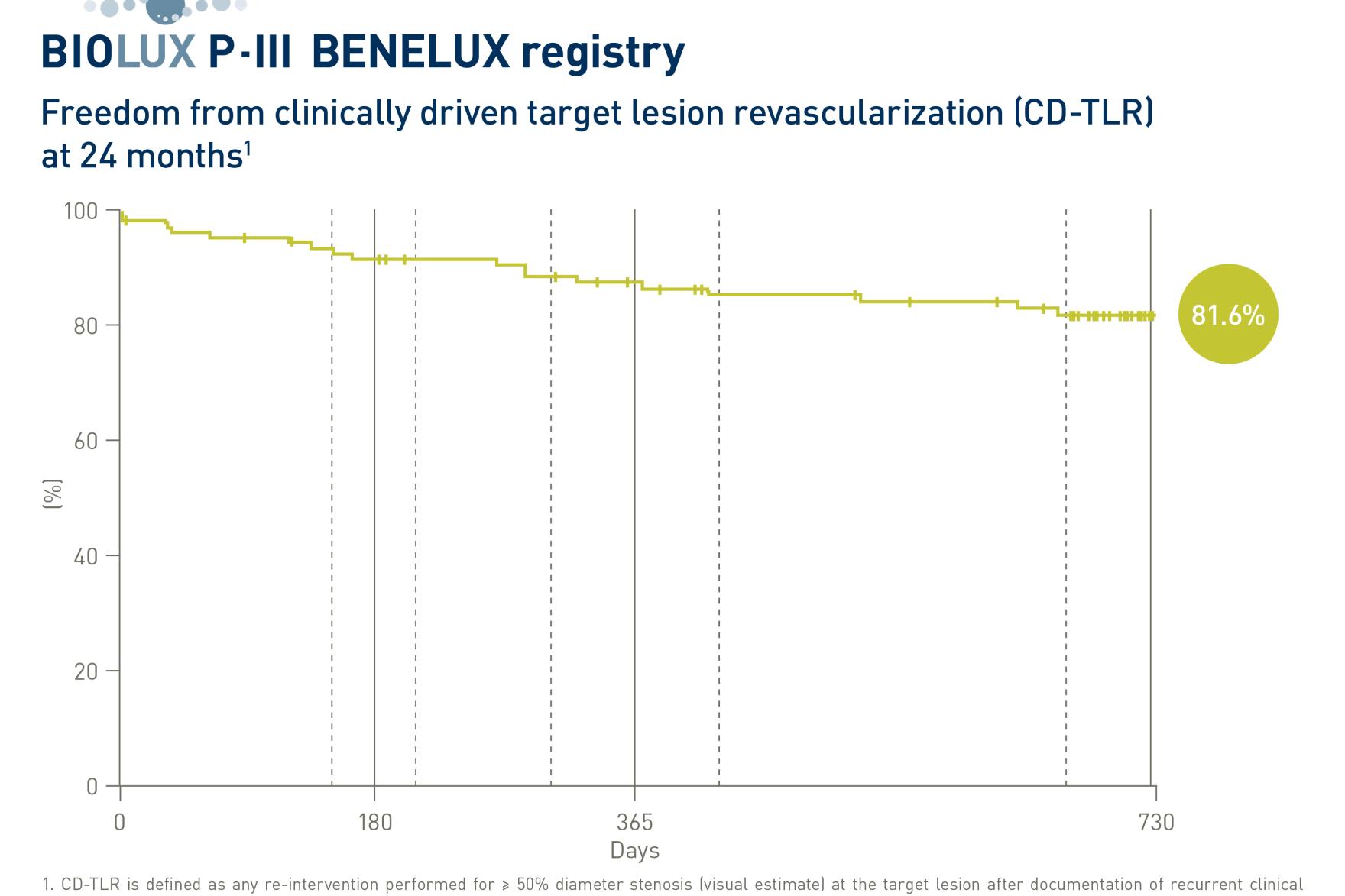
BIOTRONIK announced the two-year-results from the BIOLUX P-III BENELUX all-comers registry, presented by Principal Investigator Prof. Frank Vermassen at CIRSE 2023 in Copenhagen, Denmark.1 The prospective, international, multicenter post-market registry evaluated the safety and efficacy of the Passeo®-18 Lux® drug-coated balloon (DCB) catheter in isolated popliteal artery lesions. This indication is considered a difficult vessel bed to treat due to its biomechanical constraints that usually preclude the placement of stents.
BIOLUX P-III BENELUX registry enrolled 99 patients in Belgium, the Netherlands, and Luxembourg with Rutherford 2-5 disease and at least two centimeters of healthy vessel segment between lesions in the popliteal artery and lesions in the distal superficial femoral artery. All patients were treated with the Passeo-18 Lux DCB. The bail-out stenting rate was 14%.
At 24 months:
“We face a scarcity of data evaluating endovascular therapy for isolated popliteal artery lesions, known as a difficult vessel bed to treat as we don’t want to leave anything behind,” said Principal Investigator Prof. Frank Vermassen, Chief of Vascular and Thoracic Surgery at UZ Ghent in Ghent, Belgium. “It is encouraging to see that we can safely and effectively treat these lesions with a DCB-only approach.”
“BIOTRONIK is proud to be a leader in research of paclitaxel-coated balloons, striving to study device performance in the most challenging anatomy and patient characteristics,” said Stuart Perks, Vice President of Marketing, Vascular Intervention at BIOTRONIK. “We repeatedly see the safety and effectiveness of Passeo-18 Lux DCB demonstrated in a diverse range of clinical presentations.”
-END-
References:
1. Vermassen F. A prospective, international, multi-centre, post-market all-comers registry to assess the clinical performance of the Passeo-18 Lux DCB in isolated popliteal arteries – BIOLUX P-III BENELUX. Presented at: CIRSE 2023; September 9-13, 2023; Copenhagen, Denmark.
2. CD-TLR is defined as any re-intervention performed for ≥ 50% diameter stenosis (visual estimate) at the target lesion after documentation of recurrent clinical symptoms of the patient.
Disclaimer:
Passeo and Lux are trademarks or registered trademarks of the BIOTRONIK Group of Companies.
Passeo-18 Lux paclitaxel-releasing PTA balloon catheter is not available for sale or use in the United States .
For more information: BIOTRONIK Passeo 18 Lux - Drug-Coated Balloon Catheter
About BIOTRONIK:
At BIOTRONIK, patient well-being is our top priority and has been for 60 years. BIOTRONIK is a leading global medical technology company with products and services that save and improve the lives of millions suffering from heart and blood vessel diseases as well as chronic pain. Driven by a purpose to perfectly match technology with the human body, we are dedicated innovators who develop trusted cardiovascular, endovascular and neuromodulation solutions. BIOTRONIK is headquartered in Berlin, Germany, and is represented in over 100 countries.